News

Batch Forensics: The Case for Link Groups – Class based Unit Synchronization
To coordinate activities between units, take advantage of product functionality to greatly reduce controller code. Programing and unit coordination are used during activities such as material transfer from one unit to another and may be used to coordinate recipe pause points in a unit while the recipe of another unit reaches the desired step, or may be used to transfer process data from one unit to another for further evaluation, etc.
Using product functionality to create a class-based solution while minimizing programming greatly simplifies the complexity of the code. The product functionality consists of link groups and Phase logic phase requests (PXRQ for phase manager phases and RQ for classic OPC phases).
To better understand its usability, we will use a sample process consisting of units capable of transfer material with each other, One to Many, Many to One, Many to Many. In any of these examples, keeping track of who the groups involved in a synchronization can be as simple as specifying the required pairs (more than two concurrent can be done as well) in the procedural (recipe) model.
Let’s look at several scenarios:
Read More | Posted In: Batch Boosters, FactoryTalk Batch

Dosing Materials Based On Their Properties
Improving Product Quality
Often the quantities of materials required to manufacture a product are specified by weight or by volume. Recipes and procedures are created to specify the activities that need to be performed and the number of materials required.
The Setpoints for these quantities are set in the recipes based on the materials that are being used, if the material properties change significantly then these recipe quantities need to be updated once the new materials enter the production stream. This often requires tracking the material consumptions and updating the recipes at the appropriate time. i.e. late at night on Saturday once the new material is introduced to the production environment.
These new formula quantities are calculated after a lab analysis is performed of each Lot sample. The timing to introduce these new recipe parameter values can be critical to the Quality and the Cost of the product.
Read More | Posted In: Bakery Industry, Batch Boosters, Beverage Industry, Chemical Industry, Distilleries, Food Industry, Life Science Industry, White Papers

Material Dosing Geneology
The Food industry uses batch manufacturing extensively and therefore requires traceability for material ingredients, i.e. the ability to follow the movement of a food product and its’ constituents all through the production process.
This capability not only largely prevents unsafe foods from reaching the consumer but also improves product quality and efficiency in the event of a recall.
Read More | Posted In: Bakery Industry, Batch Boosters, Beverage Industry, Chemical Industry, Distilleries, Food Industry, Life Science Industry, White Papers
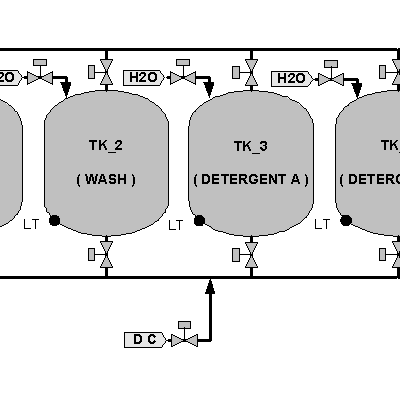
Flexible and Modular CIP and SIP Automation Enables Continuous Improvements
Clean In Place (CIP) and Steam in Place (SIP) constitute critical aspects of the bottom line of a manufacturing process, just as important as the products manufactured with the assets, CIP and SIP play a very important role in the product quality as well as the overall availability of the equipment to manufacture these products. Being able to consistently execute the required cleaning step regardless of the equipment level of automation is paramount, and being able to clearly specify and enforce the steps required are critical regardless of how these tasks are executed. Operators and equipment should seamlessly perform their task to ensure the desired cleaning procedures are executed in an optimum manner.
Read More | Posted In: Batch Boosters, Clean in Place, White Papers

Securing your Intellectual Property
Production procedures and formula values property
How secure is the intellectual property of your products?
Many degrees of automation exist in manufacturing plants, but they all have a common requirement– specifying the activities and the order in which they need to occur as well as the key setpoints such as materials, amounts, tolerances, temperatures, durations, etc.
Read More | Posted In: Bakery Industry, Batch Boosters, Beverage Industry, Chemical Industry, Distilleries, Food Industry, Life Science Industry, MES, Metals, White Papers

Improve Cooking Quality Utilizing the Lethality Factor Principles
Cooking (thermal processing) is critically important for controlling the levels of foodborne pathogens in products. Cooking is an effective process for eliminating, or at least reducing to acceptable levels, any pathogenic bacteria that may be present in the products. Today thermal processing is applied to literally hundreds of types of manufacturing processes from hams to jerky and many other industries.
There is a wide variety of processes, schedules, and equipment used in cooking areas. Furthermore, the products have unique compositions, e.g., fat and moisture content, ingredients, and other characteristics that can create different responses to the thermal processing technology used. It becomes important to appreciate how different processing systems and schedules affect product lethality, i.e., the capacity to cause death or serious harm to the non-desirable.
Read More | Posted In: Bakery Industry, Batch Boosters, Food Industry, White Papers

Materials and Lot ID Traceability
In the event of a recall can you find everything you need to know in an expeditious way.
As per Wikipedia, “Traceability” is the capability to trace something. In some cases, it is interpreted as the ability to verify the history, location, or application of an item by means of documented recorded identification.
In the process industry, we are required to know the whereabouts of the raw and intermediate materials as well as the finished products. This is paramount since recalls can be impactful to the safety of the consumer as well as financially to the producers.
Read More | Posted In: Bakery Industry, Batch Boosters, Beverage Industry, Chemical Industry, Distilleries, Food Industry, Life Science Industry, White Papers
Automated Procedures for Manual Processes
Process customers do not need to automate their equipment to start taking advantage of some of the important benefits that off-the-shelf automation products possess. Processes that are typically operated manually can benefit from automating their procedural execution, meaning that the activities that operator typically perform by following printed Standard Operating Procedures can be Specified, Enforced, and Verified in electronic form.
Off-the-shelf products allow us to Specify recipes that can span multiple unit procedures. These can be enforced by prompting the operator or operators of the different areas to perform the required manual activities in the order specified, information normally captured on a sheet of paper or batch log can be annotated and journaled electronically and then this information can be instantly used to make decisions on the active recipe (i.e. adjust the amount of material to be added based on a sample analysis).
Read More | Posted In: Bakery Industry, Batch Boosters, Beverage Industry, Chemical Industry, Distilleries, Food Industry, Life Science Industry, Metals, White Papers
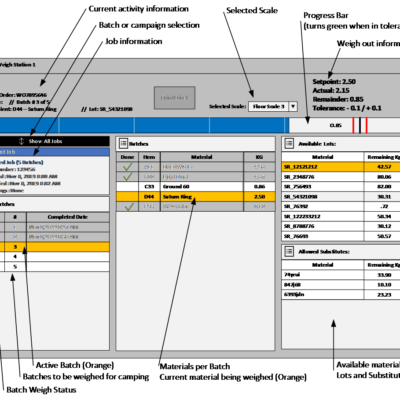
Weighing In Dispensing System
Manual weighing and dispensing tracked electronically.
Automating the dosing of Minor and Micro ingredients is often cost-prohibitive. These tasks are often performed manually by operators that weigh the required materials then dose them to the required destination at the appropriate time. These procedures are typically guided by Paper Standard Operating Procedures (SOP’s) that are used to track progress and capture required information manually.
These activities can be conducive to errors that are captured downstream after additional value has been added to the raw materials, some of these errors cause batches to be reprocessed or rejected.
Read More | Posted In: Bakery Industry, Batch Boosters, Beverage Industry, Chemical Industry, Distilleries, Food Industry, Life Science Industry, White Papers

Material Storage Management
Automation Simplified
Managing the reception of materials and its distribution throughout all the storage locations can be a daunting task. Automating this with off-the-shelf products simplifies tremendously the implementation of such a solution. Factory Talk Batch has a component called Material Manager that enables such implementation.
This solution seamlessly integrates the reception of materials into the Plant with the production process. Setting up this solution consist of defining the materials capabilities in Factory Talk Batch. These capabilities are defined by containers, or locations where material can reside, possible materials that can exist in the plant and the possible means of moving material from a source to a destination called Phases.
Read More | Posted In: Bakery Industry, Batch Boosters, Beverage Industry, Chemical Industry, Distilleries, FactoryTalk Batch, Food Industry, Life Science Industry, White Papers
